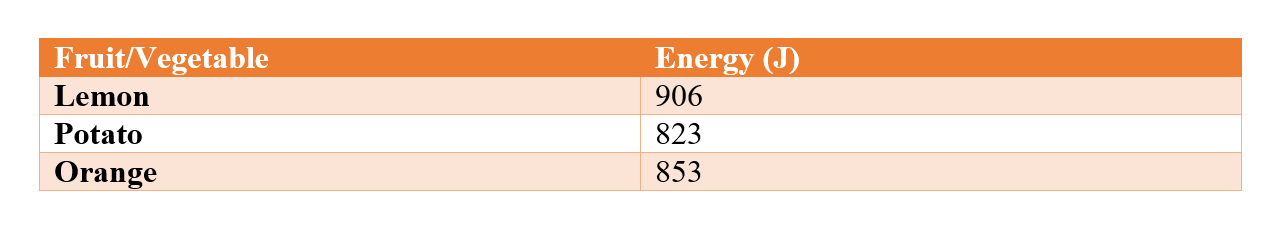Lemon Battery Experiment Aim: To make a battery using fruit and metal Keywords: Cathode: A negatively charged electrode that all electrons enter the electrical device through. Anode: A positively charged electrode through which all electrons leave the electrical device Electrolyte: Substances within the body which contain ions that help the body perform different functions such as contracting muscles and regulating the heartbeat. Focus Question: How Many Oranges does it take to Charge and IPhone? Approx. 2 380 orange slices, Approx. 595 oranges. In order to calculate the amount of oranges required to charge an IPhone, we must first look at the amount of energy required to bring the IPhone battery up to 100%. The IPhone 6, to be fully charged, requires 10.5 watt hours, which is equal to 37 800J. From our experiment, we know that the amount of energy that produced by the orange was 853J. Now, if we divide 37 800 by 853, we get approx. 44.3 oranges. This number is quite odd, as the numbers above following the focus question came from a video explaining this very question, and they suggest that we would need over 13 times the amount of oranges as the amount that I calculated. In this lesson we performed the ‘Lemon Battery Experiment’ to see whether we could make a battery out of fruit and metal. The two different metals we used were copper and zinc, which acted as our anode (positive side to the battery) and our cathode (negative side) respectively. We measured the amount of energy that flowed through a lemon, potato and an orange, and ordered them from the least energy flow to the most. How does the ‘Lemon Battery Experiment’ work? In five dot points:
From the above table you can see that the potato had the least amount of energy on 823J, with the orange in the middle with 853J, and the lemon in first, beating the orange by over 50J, on 906J. Lesson Summary
0 Comments
Leave a Reply. |
Your browser does not support viewing this document. Click here to download the document.
|